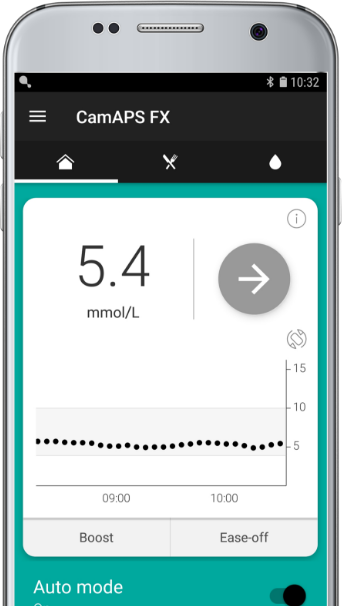
CamAPS® FX by CamDiab Ltd
CamAPS FX is the first CE marked Hybrid Closed Loop Algorithm compatible with the Dana RS Insulin Pump.
The hybrid closed-loop system that automatically adjusts insulin delivery on your insulin pump based on your sensor glucose readings.
Adaptive and personalised to you
Learns and adjusts according to your insulin needs, whilst giving you the freedom to change settings.
Runs on your smartphone
Compatible with the Android OS*.
*Android is a trademark of Google LLC
Available for all ages
Approved for ages one year and over.
Comprehensively tested
Developed at the University of Cambridge. Considered by many as ‘the best algorithm out there’. Randomised clinical trials have shown a significant increase in time in range, a reduction in HbA1c, as well as a reduction in time spent in hypoglycaemia and hyperglycaemia and improved well-being.
- https://doi.org/10.1016/S0140-6736(18)31947-0
- https://doi.org/10.1056/NEJMoa1509351
- https://doi.org/10.1056/NEJMoa1602494
- https://doi.org/10.1056/10.2337/dc18-1881
- https://doi.org/10.1177/1932296817702656
- https://diabetes.medicinematters.com/artificial-pancreas-systems/type-1-diabetes/the-artificial-pancreas–what-s-on-the-horizon-/16939360
What is CamAPS FX?
CamAPS FX is an Android app to manage glucose levels in people with type 1 diabetes, aged one year and older, using a hybrid closed-loop approach.
Additional age restrictions may apply depending on your chosen continuous glucose monitor or insulin pump.
CamAPS® FX is a registered trademark of CamDiab Ltd.
CamAPS FX vs Sensor Augmented Pump Therapy.
The Tauschmann et al study ‘Closed-loop insulin delivery in suboptimally controlled type 1 diabetes: a multicentre, 12-week randomised trial’ published in the Lancet 2018; 392: 1321–29 compares the use of the CamAPS FX hybrid closed loop system to sensor augmented pump therapy.
It is important to note that the comparative system is sensor augmented pump therapy, rather than standard pump therapy. It is well documented that insulin pump therapy health benefits are already improved with sensor augmentation over standard insulin pump therapy where blood glucose data is provided by finger sticks tests at certain points in a users day.
The study is available to read in full here: http://dx.doi.org/10.1016/S0140-6736(18)31947-0
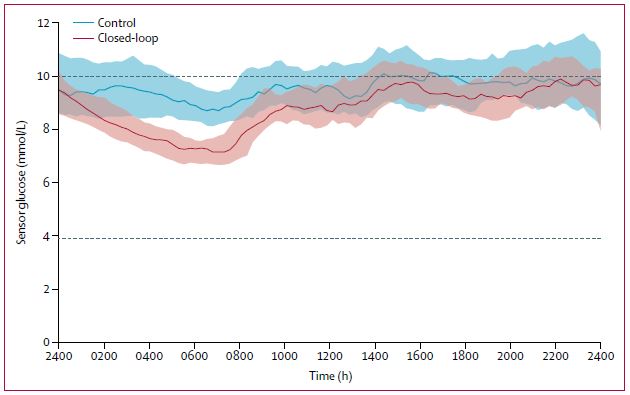
Over 12 weeks of free living, subjects were randomly provided either the CamAPS FX hybrid closed loop system or sensor augmented pump therapy. Subjects were over the age of 6 years old with sub-optimally controlled blood glucose. The primary endpoint was the proportion of time that glucose concentration was within the target range of 3·9–10·0 mmol/L at 12 weeks post randomisation.
Results:
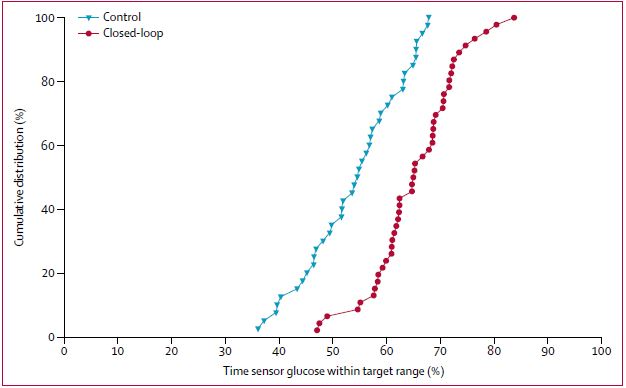
The proportion of time that glucose concentration was within the target range was significantly higher in the closed-loop group (65%, SD 8) compared with the control group (54%, SD 9).
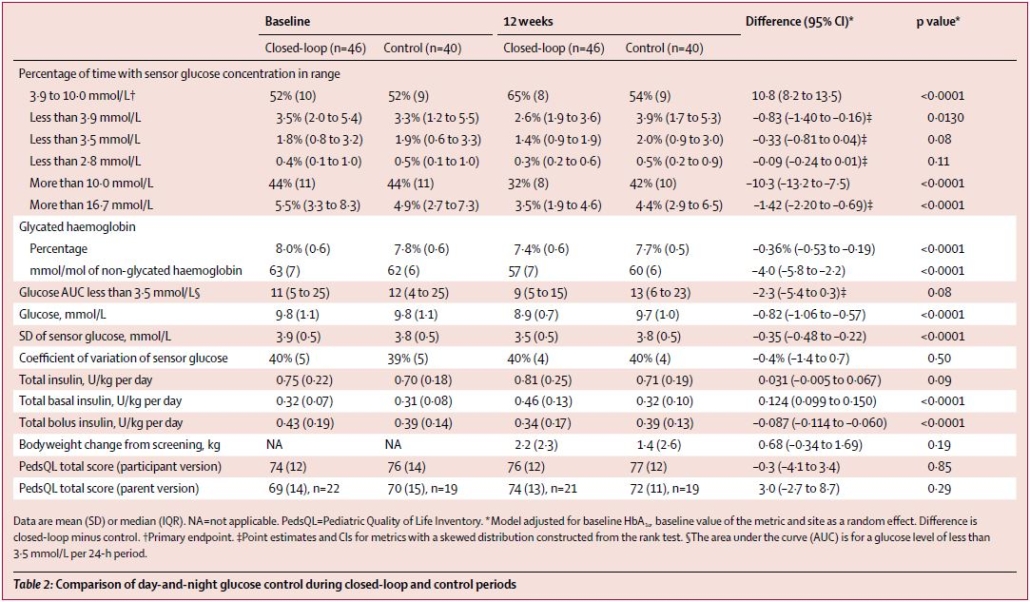
In the closed-loop group, HbA1c was reduced from a screening value of 8·3% (SD 0·6) to 7·4% (SD 0·6) after the 12-week intervention period. In the control group, the HbA1c values were 8·2% (SD 0·5) at screening and 7·7% (SD 0·5) after intervention; reductions in HbA1c percentages were significantly greater in the closed-loop group compared with the control group.
The time spent with glucose concentrations below 3·9 mmol/L (mean difference in change –0·83 percentage points, –1·40 to –0·16; p=0·0013) and above 10·0 mmol/L (mean difference in change 10·3 percentage points, –13·2 to –7·5; p<0·0001) was shorter in the closed-loop group than the control group.
Interpretation:
Hybrid closed-loop insulin delivery improves glucose control while reducing the risk of hypoglycaemia across a wide age range in patients with suboptimally controlled type 1 diabetes.