Insulin Pump Therapy:
Diabetes in toddlers & preschool children with type 1 DM
Litton et al 2002 (n=9 aged 10-40 months)
Factors leading to poor control
- Unpredictable food intake
- Unpredictable activity levels
- Imprecise administration of low doses of insulin
- Frequent viral infections
- Inability to communicate symptoms of low BG, predisposing to frequent hypos
Diabetes in toddlers & preschool children with type 1 DM
Litton et al 2002 (2)
- Patient selection criteria
- Recurrent moderate/severe hyperglycaemia
- HbA1c persistently > 9.0
| Pre-pump | Post-pump | |
| 9.5 +/- 0.4 | 7.9 +/- 0.3 | |
| Insulin dose | no sig diffs | |
| BG recordings | no sig diffs | |
| Linear growth | no sig diffs | |
| Weight gain | no sig diffs | |
| Severe hypos/ mth | 0.52 +/- 0.1 | 0.09 +/_ 0.02 |
| Parental clinic contact-day | 5.9 +/- 1.5 | 46.3 +/- 4.4 |
Diabetes in toddlers & preschool children with type 1 DM
Litton et al 2002 (4)
- A controlled study of the effects of discontinuation of pump therapy could not be conducted because all our families preferred CSII to MDI and refused to consider a return to previous modes of insulin administration
- …the frequency of parental contacts with health personnel declined by >80 continued with CSII
Pumps & Paediatrics
Aherne et al 2002 (n=169: age range=1.5-18 yrs)
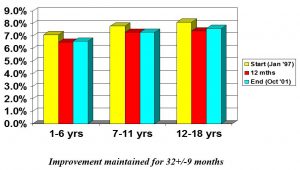
- “CSII [insulin pump therapy] is an effective alternative to injection therapy in a large paediatric diabetes clinic setting. Even very young patients can utilise CSII to safely lower HbA1c levels”.
- “Improved diabetes control was achieved with CSII without increasing daily insulin doses and in association with a decrease in the frequency of severe hypoglycaemic events (p=0.05 vs prepump, all three ages combined)”.
- “There was a significant and consistent reduction in mean HbA1c levels after 12 months of CSII…(p=<0.02 vs prepump) that was maintained at the most recent visit”.
- “The remarkable effectiveness of CSII in our youngest patients indicates that the child’s age should not be a barrier to the initiation of this therapy”
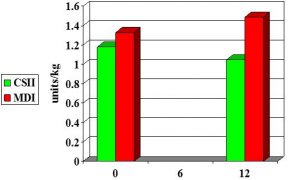
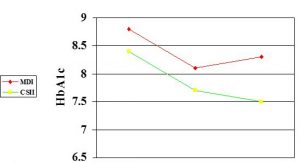
CSII vs MDI in adolescents: n=75
Boland et al 2000
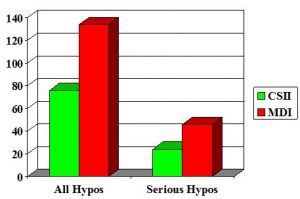
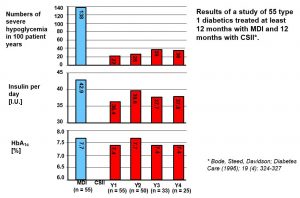
Severe hypos – events/100 pt yrs (n=55)
Bode et al 1996
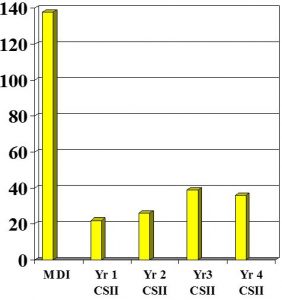
- Severe hypos 138 (MDI) vs 22 (Yr 1 CSII).. 36 (Yr 4 CSII)
- Total insulin dose reduced from 42.9 (MDI) to 36.4 (Yr1)..37.8 (Yr4)
- DKA rates were 14.6 / MDI vs 7.6 CSII per 100 pt years ie – No significant difference
DKA Rates in Children & Adolescents (n=6)
Steindel et al 1995
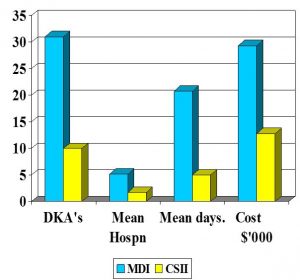
- MDI vs CSII
- DKA episodes 31 vs 10
- Mean no. hospitalisation/pt 5.2 vs1.7
- Mean no. hospital days/pt 20.8 vs 5
- Mean Treatment costs/pt – $’000 $29.33 vs $12.76
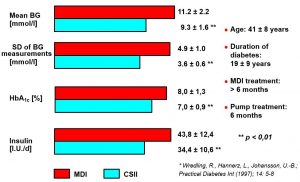
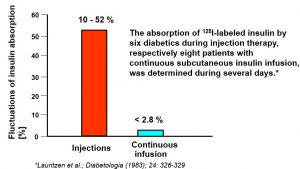
Less Day-to-day Fluctuations of Insulin Absorption During CSII Compared to Single Injections of Intermediate-acting Insulin
The absorption of 125I-labeled insulin by six diabetics during injection therapy, respectively eight patients with continuous subcutaneous insulin infusion, was determined during several days.*
Blood Glucose, HbA1c and Insulin Requirement of 14 Type 1 Diabetics Under CSII Compared to MDI Pre-treatment*
Blood glucose, HbA1c and insulin requirement under MDI and CSII
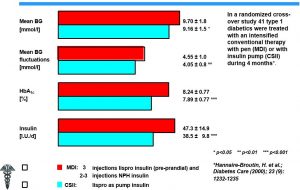
Severe Hypoglycemia, Insulin Requirement and HbA1c During MDI and CSII
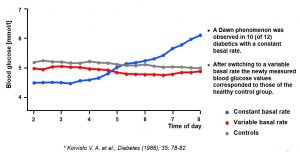
Compensating Dawn Phenomenon With Continuous Subcutaneous Insulin Infusion (CSII) With Variable Basal Rate
Blood glucose development between 2 and 8 am in 12 C-peptide negative type 1 diabetics (mean age: 30 ± 2 years; mean diabetes duration: 11 ± 2 years; HbA1: 8.9 ± 0.3 ) under continuous insulin infusion compared to 8 healthy probands
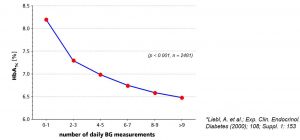
Correlation Between HbA1c and Frequency of Blood Glucose Self-monitoring Under Insulin Pump Treatment
Results of a questionnaire* on diabetics with an insulin pump treatment in Germany in 1998/1999. 2481 data sets
{(Diabetes duration: 20 ± 11 years; Treatment with an insulin pump: 3 ± 5 years; Frequency of daily blood glucose self-monitoring: 5.4 ± 1.8; HbA1c: 6.9 ± 1.1 ; standard range: 6 – 9 )
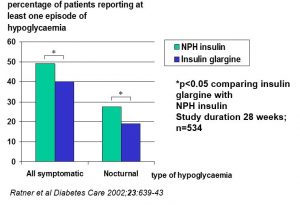
Effect of insulin Glargine compared with NPH on the frequency of hypoglycemia in patients with type 1 diabetes
References
1. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. New England Journal of Medicine 1993;329:977-986.
2. The Diabetes Control and Complications Trial Research Group. Implementation of treatment protocols in the Diabetes Control and Complications Trial. Diabetes Care 1995;18(3):361-76.
3. Lauritzen T, Faber OK, Binder C. Variation in 125I-insulin absorption and blood glucose concentration. Diabetologia 1979;17(5):291-5.
5. Lauritzen T, Pramming S, Gale EA, Deckert T, Binder C. Absorption of isophane (NPH) insulin and its clinical implications. British Medical Journal (Clinical Research ed.) 1982;285(6336):159-62.
5. Binder C, Lauritzen T, Faber O, Pramming S. Insulin pharmacokinetics. Diabetes Care 1984;7(2):188-99.
6. Lauritzen T, Pramming S, Deckert T, Binder C. Pharmacokinetics of continuous subcutaneous insulin infusion. Diabetologia 1983;24(5):326-9.
7. Geffner ME, Frank HJ, Kaplan SA, Lippe BM, Levin SR. Early-morning hyperglycemia in diabetic individuals treated with continuous subcutaneous insulin infusion. Diabetes Care 1983;6(2):135-9.
8. Koivisto VA, Yki-Jarvinen H, Helve E, Karonen SL, Pelkonen R. Pathogenesis and prevention of the dawn phenomenon in diabetic patients treated with CSII. Diabetes 1986;35(1):78-82.
9. The Diabetes Control and Complications Trial Research Group. Adverse events and their association with treatment regimens in the diabetes control and complications trial. Diabetes Care 1995;18(11):1415-27.
10. Eichner HL, Selam JL, Holleman CB, Worcester BR, Turner DS, Charles MA. Reduction of severe hypoglycemic events in type I (insulin dependent) diabetic patients using continuous subcutaneous insulin infusion. Diabetes Research 1988;8(4):189-93.
11. Bode BW, Steed RD, Davidson PC. Reduction in severe hypoglycemia with long-term continuous subcutaneous insulin infusion in type I diabetes. Diabetes Care 1996;19(4):324-7.
12. Haardt MJ, Berne C, Dorange C, Slama G, Selam JL. Efficacy and indications of CSII revisited: the Hotel-Dieu cohort [letter]. Diabetic Medicine 1997;14(5):407-8.
13. Tsui EY, Chiasson JL, Tildesley H, et al. Counterregulatory hormone responses after long-term continuous subcutaneous insulin infusion with lispro insulin. Diabetes Care 1998;21(1):93-6.
14. Kanc K, Janssen MM, Keulen ET, et al. Substitution of night-time continuous subcutaneous insulin infusion therapy for bedtime NPH insulin in a multiple injection regimen improves counterregulatory hormonal responses and warning symptoms of hypoglycaemia in IDDM. Diabetologia 1998;41(3):322-9.
15. Launay B, Zinman B, Tildesley HD, Strack T, Chiasson JL. Effect of continuous subcutaneous insulin infusion with lispro on hepatic responsiveness to glucagon in type 1 diabetes. Diabetes Care 1998;21(10):1627-31.
16. Jovanovic L, Druzin M, Peterson CM. Effect of euglycemia on the outcome of pregnancy in insulin-dependent diabetic women as compared with normal control subjects. American Journal of Medicine 1981;71(6):921-7.
17. Kitzmiller JL, Gavin LA, Gin GD, Jovanovic-Peterson L, Main EK, Zigrang WD. Preconception care of diabetes. Glycemic control prevents congenital anomalies. JAMA 1991;265(6):731-6.
18. Rudolf MC, Coustan DR, Sherwin RS, et al. Efficacy of the insulin pump in the home treatment of pregnant diabetics. Diabetes 1981;30(11):891-5.
19. Potter JM, Reckless JP, Cullen DR. The effect of continuous subcutaneous insulin infusion and conventional insulin regimes on 24-hour variations of blood glucose and intermediary metabolites in the third trimester of diabetic pregnancy. Diabetologia 1981;21(6):534-9.
20. Kitzmiller JL, Younger MD, Hare JW, et al. Continuous subcutaneous insulin therapy during early pregnancy. Obstetrics and Gynecology 1985;66(5):606-11.
21. Guerci B, Meyer L, Delbachian I, Kolopp M, Ziegler O, Drouin P. Blood glucose control on Sunday in IDDM patients: intensified conventional insulin therapy versus continuous subcutaneous insulin infusion. Diabetes Research and Clinical Practice 1998;40(3):175-80.
22. Ronn B, Mathiesen ER, Vang L, Lorup B, Deckert T. Evaluation of insulin pump treatment under routine conditions. Diabetes Research and Clinical Practice 1987;3(4):191-6.
23. Wolf FM, Jacober SJ, Wolf LL, Cornell RG, Floyd JCJ. Quality of life activities associated with adherence to insulin infusion pump therapy in the treatment of insulin dependent diabetes mellitus. Journal of Clinical Epidemiology 1989;42(12):1129-36.
24. Boland EA, Grey M, Oesterle A, Fredrickson L, Tamborlane WV. Continuous subcutaneous insulin infusion. A new way to lower risk of severe hypoglycemia, improve metabolic control, and enhance coping in adolescents with type 1 diabetes. Diabetes Care 1999;22(11):1779-84.